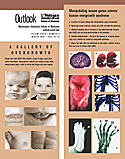
Understanding Overgrowth
Additional or overgrown body parts are hallmarks of little-understood Simpson Golabi Behmel Syndrome.
A collaborative research effort examines this rare condition from two different perspectives.
|
Learn some of the signs of SGBS and see how researchers are manipulating mouse genes to replicate these characteristics. PDF FORMAT 96 K |

Linked to the X chromosome, SGBS occurs predominantly in males.

“Most pediatricians have never heard of SGBS, let alone seen a child with the syndrome. No one knows what the risks are for the various cancers, or even the spectrum of medical problems that occur in SGBS patients.”
MICHAEL R. DEBAUN, MD, MPH


![]()
EARLY IN THEIR CAREERS, physician-scientists typically choose a disease to study. For Scott Saunders, MD, PhD, assistant professor of pediatrics and of molecular biology and pharmacology, however, the disease found him. It happened when another School of Medicine scientist linked a mutated gene to a rare congenital condition known as Simpson Golabi Behmel Syndrome (SGBS).
That finding redirected Saunders’ research and eventually
led to a fruitful collaboration with Michael R. DeBaun, MD, MPH, assistant
professor of pediatrics, and to the establishment of the first clinic
and registry for families coping with SGBS.
Included in that group are Patti and Robert Valentine of O’Fallon
IL, whose son Justin was born in 1991—five weeks premature, but weighing
8 lbs, 7oz. The baby couldn’t eat or swallow well, his sucking reflex
was poor, and his overall muscle tone was weak.
The root of his problems was a mystery. After Justin’s birth, Patti, a former nurse, stood with her newborn son’s pediatrician at the hospital nurse’s station paging through medical books trying to identify his condition. Nothing quite fit.
The pediatrician referred Patti and her husband, then an Air Force lieutenant stationed in Great Falls MT, to a medical geneticist in Helena. He was able to diagnose Justin with Simpson Golabi Behmel Syndrome (SGBS), one of several so-called overgrowth syndromes that are characterized in part by babies with large head and body size.

Michael R. DeBaun, MD, MPH, and Scott Saunders, MD, PhD
Linked to the X chromosome, SGBS occurs predominantly in males. First described in 1975, half of children with SGBS die at birth, probably due to heart defects. Those who survive can have a range of conditions including an enlarged tongue, widely spaced eyes, cleft palate, bone abnormalities such as extra or fused fingers and toes, and organ abnormalities that can involve the heart, kidneys, bladder and intestines. In addition, children with SGBS are at high risk for three childhood cancers: Wilm’s tumor, which affects the kidneys, neuroblastoma and liver cancer.
Patti left the geneticist’s office with Justin’s diagnosis and a thin folder of information containing most of what was then known about SGBS. Over the next nine years, the family was transferred three times. In each new location, Patti had to find new doctors for Justin and often found herself educating them about the obscure syndrome.
In 1999, she found DeBaun. While searching the Internet for information, she found a web page devoted to another overgrowth condition, Beckwith Wiedemann Syndrome. The site mentioned SGBS and included a picture of DeBaun.
“He looked like a nice guy, so I e-mailed him,” Patti recalls. To her surprise, DeBaun replied within minutes—he had been checking his e-mail when her message arrived. He informed her about the Washington University SGBS clinic and registry at St. Louis Children’s Hospital. A few months later, the Valentine family attended the clinic.
The Saunders/DeBaun collaboration began in the late 1980s, when Saunders was studying a group of little-understood molecules known as heparan sulfate proteoglycans (HSPGs). HSPGs, which consist of proteins with sugars attached to them, are now known to be essential for guiding development and growth of bones and organs in embryos.
In 1997, Saunders joined the faculty at Washington University. The year before, David Schlessinger, PhD, then a researcher in the Department of Genetics, had discovered a mutation on the human X chromosome that linked an HSPG gene to Simpson Golabi Behmel Syndrome. The gene, known as glypican 3 (gpc3), was related to the HSPG genes that Saunders had been studying. When he learned about Schlessinger’s discovery, he refocused his research on the gpc3 gene and its relation to SGBS.
“Physician-scientists like myself rarely have the opportunity to see our research directly help the types of patients we care for,” says Saunders. “HSPGs were the focus of my laboratory research, so when I learned that gpc3 caused birth defects in children—the focus of my clinical practice—I had to change directions.”
To investigate how the gpc3 mutation causes SGBS, Saunders first engineered a strain of mice that was missing the gpc3 gene. The mouse model showed many of the same features as SGBS.
As it turned out, Saunders’ laboratory was only about 50 feet from DeBaun’s on the 11th floor of St. Louis Children’s Hospital, and a collaboration was begun.
DeBaun is a pediatric oncologist and epidemiologist interested in syndromes that predispose children to cancer, such as Beckwith Wiedemann and SGBS. He’d started a Beckwith Wiedemann registry six years earlier, and the experience equipped him to efficiently organize an SGBS registry. Saunders, a neonatologist with expertise in birth defects and molecular biology, had knowledge that could help reveal the biological mechanism behind the cancer-causing syndrome.
The two investigators held their first yearly multidisciplinary clinic in 2000, after the SGBS registry was up and running. Families have come to the clinic from as far away as Canada and Argentina. Some have attended all three years because they find it helpful to meet other children with SGBS and to share experiences.
“Most pediatricians have never heard of SGBS, let alone seen a child with the syndrome,” says DeBaun. “No one knows what the risks are for the various cancers, or even the spectrum of medical problems that occur in SGBS patients.”
In time, Saunders and DeBaun hope to answer these questions through the yearly clinic and the registry. “By keeping in touch with the families and their pediatricians,” says Saunders, “we will develop experience about the kinds of things we need to watch for.”
Saunders and DeBaun also have developed a test that confirms which patients have the gpc3 mutation, which they believe is present in 30 to 40 percent of SGBS patients. But they believe other mutations also must be involved because children with the syndrome show a range of severities.
“Why are some children more likely to develop cancer than others, and can we do anything to prevent those cancers?” asks DeBaun. “Such questions require years, if not decades, to answer. This is just the first step of a long process.”
Ultimately, the researchers hope to develop a genetic test that will determine which children will have a more or less severe form of SGBS. Hundreds of patients are needed for such analyses, and after three years, the registry includes just 17 families. “That is far more than anyone else has seen,” says DeBaun, “but we need more to learn what we’d like to learn.”
Fortunately, important clues about SGBS also will come from Saunders’ mice. Studies of the mice, for example, will help to identify the other genes and mutations that are believed to influence the severity and course of the syndrome.
Already, study of the mice has provided insight into changes that might occur in human patients. For example, research by others has suggested that the mice might undergo immune-system changes that make them susceptible to certain infections. “If we can verify that similar changes do occur in humans,” says Saunders, “we can help patients and their families by telling them that they may be at risk for certain infections.”
And patients and families can provide researchers with clues about changes they should look for in the mice. During this year’s SGBS clinic, one mother commented that her son and the other children in the waiting room all seemed to have knobby knees. A postdoctoral student in Saunders’ lab had a short time earlier reported that the knees of the mice looked overly broad.
“That mother’s casual observation made us rethink
some of the studies we were doing and may help us ask better questions
about what is happening in the bones of the mice,” says Saunders.
“As we learn more, we share it in the scientific literature. This
helps other geneticists, who may have an infant in the neonatal intensive
care unit, as they try to determine if their patient has SGBS.” ![]()
![]()