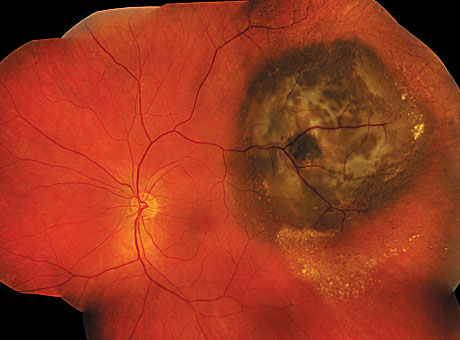
A cancerous melanoma tumor (dark area, upper right) is seen below the retina. HARBOUR LABORATORY

A cancerous melanoma tumor (dark area, upper right) is seen below the retina. HARBOUR LABORATORY
Washington University in St. Louis has licensed to Castle Biosciences Inc. the exclusive use of a gene to detect the spread of cancer in melanoma patients.
A link between the BAP1 gene and cancer metastasis was discovered by Washington University scientists J. William Harbour, MD, an ophthalmologic oncologist, and Anne M. Bowcock, PhD, a geneticist. Their research, published in 2010 in the journal Science, showed that BAP1 mutations occurred in 84 percent of melanomas that developed in the eye and later spread, but were rare in eye tumors that did not metastasize.
The researchers also have identified one individual with a germline BAP1 mutation, indicating that this mutation can be inherited and give rise to a familial cancer syndrome. Mutations in the BAP1 gene have been found in skin melanomas, mesotheliomas, menin-giomas and cancers of the breast, ovary, kidney and lung.
Harbour, the Paul A. Cibis Distinguished Professor of Ophthalmology and Visual Sciences, and Bowcock, professor of genetics, pediatrics and medicine, worked closely with the university’s Office of Technology Management, which filed patents related to the BAP1 discovery, giving the university the option to license the technology.
Mutations in the BAP1 gene have been found in skin melanomas, mesotheliomas, meningiomas and cancers of the breast, ovary, kidney and lung.
“This is a win-win situation for the university,” says Bradley J. Castanho, PhD, director of the university’s Office of Technology Management. “Licensing the technology maximizes the opportunities for WUSTL scientists and the university to take this discovery to the market, where it could be developed into a diagnostic test for cancer metastasis.”
Castle Biosciences, based in Texas, is validating the BAP1 technology for use in both ocular melanoma and melanoma that develops on the skin. The company plans to incorporate BAP1 as a target in a clinical test for melanoma metastasis.
The new license is the second between the university and Castle Biosciences for technology developed by Harbour. The first exclusive license, in 2009, was related to technology to predict the risk of metastasis in patients with ocular melanoma. The company has completed technical and clinical validation studies, and the assay is now part of the standard of care for patients with the disease.