What we can do for
CANTU syndrome
The world’s only clinic explores the science behind a rare syndrome
By Michael C. Purdy
Learn More
The Center for Investigation of Membrane Excitability Diseases (CIMED)
FOR SEVEN YEARS, Randall and Rachel Lamfers struggled to get a diagnosis for their son, Noah, who was born prematurely, had problems with pulmonary hypertension and needed a breathing tube. In November 2013, analysis of Noah’s DNA finally gave them a diagnosis: a rare condition called Cantu syndrome.
An Internet search quickly led them to Washington University for treatment. This summer, the Lamfers, who live in Sioux Falls, S.D., brought Noah to St. Louis.
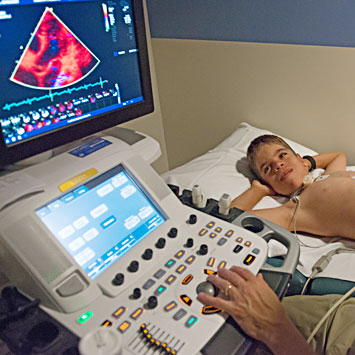
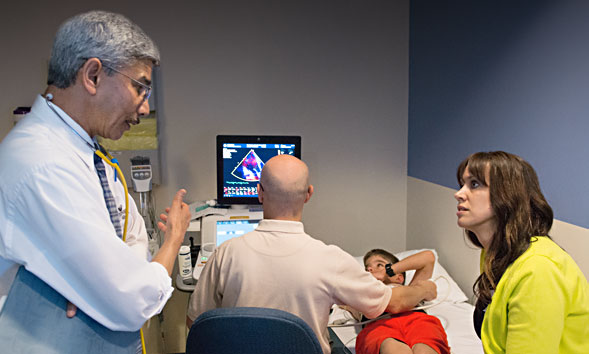
In June, researchers and physicians at the School of Medicine and St. Louis Children’s Hospital hosted the world’s only medical clinic for patients with Cantu syndrome, providing some treatment options and support for families, who, until this time, may have never met others with the disorder. A scientific symposium was held in conjunction with the clinic.
Medical literature reports only about 50 people with Cantu, but many more now are being identified by genetic analysis, and the researchers already are following six patients in the St. Louis area, indicating that the syndrome may be significantly more common than first realized.
Cantu syndrome causes excessive hair growth, bone abnormalities, swelling in the legs, problems with heart function and a range of other symptoms that researchers are just beginning to quantify.
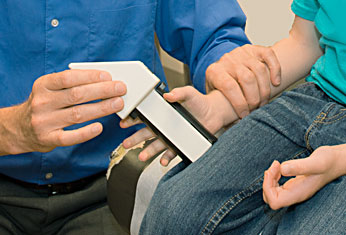
A major focus of the Cantu syndrome clinic has been in-depth study of the cardiovascular abnormalities associated with the condition.
This June marked the second time the clinic was held at the St. Louis campus. Dorothy K. Grange, MD, professor of pediatrics in the Division of Genetics and Genomic Medicine, created a registry for Cantu syndrome patients; in August 2013, she organized the first clinic, along with professor Gautam K. Singh, MD, instructor Mark D. Levin, MD, and assistant professor Beth A. Kozel, MD, PhD, all in the Department of Pediatrics.
“We’ve been waiting a long time for this,” Rachel Lamfers said. “We’re excited to be able to introduce Noah to other children with Cantu syndrome, and we want to hear the stories of other families who have children affected by this condition.”
At the first clinic, Tracy Buerck-Eggemeyer’s son, Cole Eggemeyer, then 12, was fearful of having his blood drawn.
“He asked me, ‘Mom, is having my blood drawn going to help other people who have Cantu syndrome in the future?’” recalled Buerck-Eggemeyer. “When I told him yes, he said, ‘I want to do it.’”
Bench-to-bedside link
The Cantu clinic and collaborative research efforts came about when roads coverged for two different groups at Washington University Medical Center.
At St. Louis Children’s Hospital, Grange and Singh, professor in the Division of Pediatric Cardiology, were treating a family with multiple cases of Cantu syndrome. The researchers were the first to suggest that Cantu is caused by a defect in a potassium channel gene that affects the cardiovascular system.
Meanwhile, Colin Nichols, PhD, the Carl Cori Professor in cell biology and physiology and director of the Center for Investigation of Membrane Excitability Diseases (CIMED), had linked potassium channels — openings in the surfaces of cells that let in charged potassium particles — to neonatal diabetes. To understand the role of a related channel, he had mutated the underlying gene in the cardiovascular system in mice.
In late 2012, the two groups realized that their investigations were linked. The CIMED scientists discovered their mice models with mutated potassium channels had Cantu-like symptoms. At about the same time, the clinicians’ genetic studies, conducted in collaboration with a group of Dutch researchers, led them to a gene called ABCC9.
“That’s not the same gene we were studying; we had mutated a gene called KCNJ8 in our mice,” Nichols said. “But we knew that proteins from each of these two genes come together to form a single potassium channel, and we now know that mutations in either gene can cause Cantu syndrome.”
Due to these mutations, the potassium channels remain open, making it harder for blood vessels to contract, a step that helps the circulatory system increase blood pressure when needed.
CIMED, a BioMed 21 interdisciplinary research center, specializes in studying these kinds of changes. Problems with these channels can lead to neurological and psychological disorders, cardiac arrhythmias, cystic fibrosis, diabetes and myriad other conditions. Nichols and the members of CIMED — which now include Grange and Singh — focus on improving therapies for these diseases.
Moving forward
Nichols said he believes the Cantu group is on the verge of significantly alleviating the marked lifestyle and clinical problems from which these patients suffer, and is gearing up for trials of a targeted drug therapy.
“Until now, Cantu syndrome patients have been treated solely on the basis of their symptoms, and what we’re learning now about the actual causes of those symptoms suggests that there may be better ways to treat them,” Nichols said.
“For example, even before the link to Cantu syndrome was identified, we were finding evidence that the mutation we were studying in mice causes blood vessels to relax,” Nichols added. “That can explain why the ductus arteriosus, a vessel that redirects blood flow during fetal life, fails to close normally after birth in many Cantu patients. It may also help us find new ways to close off this blood vessel in other situations.”
Based on what they’ve learned from studying Cantu syndrome patients, Washington University researchers now know more about how drugs such as Rogaine or minoxidil stimulate hair growth.
“Because of the gene mutation they carry, these patients are experiencing the equivalent of a chronic overdose of minoxidil,” Nichols said.
The clinic’s discoveries may not only help develop appropriate treatments for the cardiovascular problems and excess hair growth seen in Cantu and in other conditions, but also aid in the search for improved drugs that promote hair growth.
“The advances in our knowledge about Cantu syndrome have been incredible in the last two years,” Grange said. “I am optimistic we will have effective therapy for our patients in the near future.”